April 9, 2021
9 April 2021: Zina Kobbi CEO of AREMA will be the Keynote Speaker on the virtual conference “United Conference of Generic Medicine, Vaccines and Biosimilars” to […]
September 1, 2020
New guideline of bioequivalence was recently implemented in Morocco by the drug agency, Ministry of Health the link to the document: http://dmp.sante.gov.ma/upload/uploadinterne/Lignes%20directrices%20bioequivalence%20Maroc.pdf
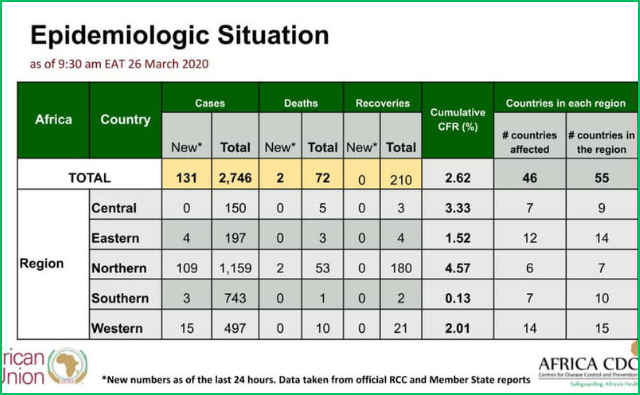
March 26, 2020
June 1, 2019
May 30, 2019
The partnership between Europe and developing countries for clinical trials (EDCTP) is providing funding of 10 million euros over five years for the “WANECAM 2” project. […]
May 3, 2019
March 26, 2019
January 2, 2019
January 1, 2019
October 9, 2018
July 29, 2018
05 July 2018: WHO launched a pilot project for prequalifying biotherapeutic medicines, a step towards making some of the most expensive treatments for cancer more widely […]
June 6, 2018
June 4, 2018
04 June 2018: FDA approves first biosimilar to Neulasta (pegfilgrastim) of Mylan GmbH. This porduct helps to reduce the risk of infection during cancer treatment . […]
April 24, 2018